2015 Fall Reports of the Commissioner of the Environment and Sustainable Development Report 1—Pesticide Safety
2015 Fall Reports of the Commissioner of the Environment and Sustainable Development Report 1—Pesticide Safety
Table of Contents
- Introduction
- Findings, Recommendations, and Responses
- Conclusion
- About the Audit
- List of Recommendations
- Exhibits:
- 1.1—Regulatory process for pesticides
- 1.2—Length of time pest control products have been conditionally registered
- 1.3—The Pest Management Regulatory Agency has allowed the use of neonicotinoids since 1995 without having confirmed whether the risks are acceptable
- 1.4—The re-evaluation of chlorpyrifos was initiated in 1999 but remains incomplete
- 1.5—The Pest Management Regulatory Agency has not kept pace with its workload of re-evaluations
- 1.6—The Pest Management Regulatory Agency took 11 years to cancel the registration of a pesticide after finding its risks unacceptable
- 1.7—The Pest Management Regulatory Agency allowed lengthy phase-out periods for some pesticides
Introduction
Background
Acceptable risk—Risk for which there is reasonable certainty that no harm to human health, future generations or the environment will result from exposure to or use of the product, taking into account its conditions or proposed conditions of registration.
Source: Pest Control Products Act
1.1 Health Canada’s Pest Management Regulatory Agency (the Agency) is the organization responsible for regulating pesticides in Canada. The Agency determines which pesticides can be sold in Canada and under what conditions they can be used. According to the Pest Control Products Act, the Agency’s primary objective is to prevent unacceptable risks to people and the environment from the use of pesticides. To accomplish this objective, the Agency evaluates proposals for new pesticides and registers those with acceptable risk for use in Canada. It also re-evaluates older pesticides against current health and environmental standards, and when alerted to potential problems, it may conduct special reviews to verify the continued acceptability of registered pesticides. When a pesticide is found to have unacceptable risks, the Agency is responsible for ensuring that it is removed from the market.
Active ingredient—The ingredient of a pesticide that controls the targeted pest. The active ingredient is combined with other ingredients to make a pesticide, or pest control product.
1.2 Approximately 7,000 pest control products, which contain about 600 active ingredients, are registered for use in Canada. Pesticides contribute significantly to agricultural productivity by preventing crop damage and to improved public health by controlling disease-carrying pests. Pesticides are used in the production of many foods Canadians eat. They are also used in gardens and homes to control weeds, insects, and rodents, and are used on pets to control parasites. Pesticides include herbicides, insecticides, and fungicides, which target weeds, insects, and fungi.

Pesticide applied to a farm field
Photo: Fotokostic/Shutterstock.com
1.3 Pesticide use is increasing. According to Statistics Canada, the area of farmland treated with herbicides, insecticides, and fungicides increased by 3 percent, 42 percent, and 114 percent, respectively, between 2001 and 2011.
1.4 Most pesticides are designed to be toxic to pests, and if used improperly, they can have serious consequences for human health and the environment. Because of these risks, pest control products are regulated (Exhibit 1.1). When the Pest Management Regulatory Agency determines that the risks posed by a pesticide are unacceptable, it may deny, amend, or cancel the registration of that product.
Exhibit 1.1—Regulatory process for pesticides
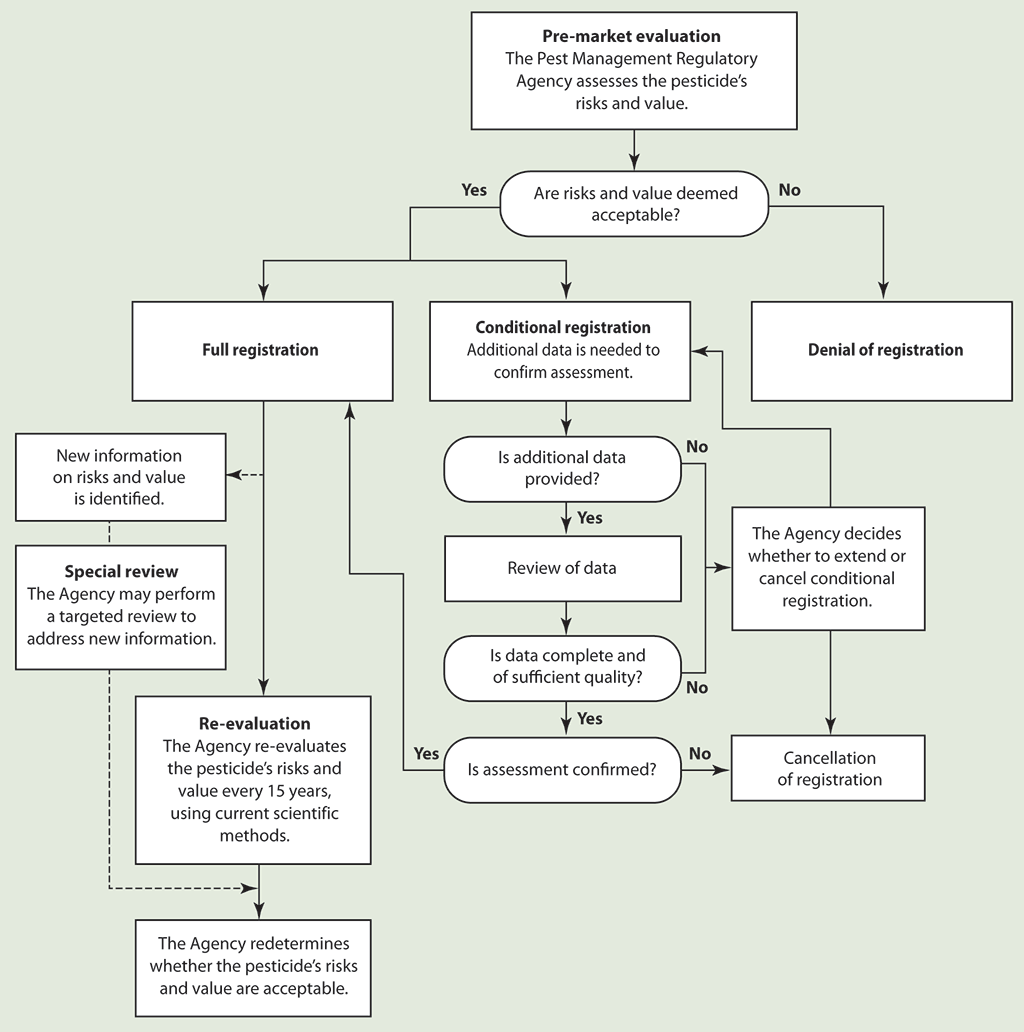
Source: Based on information provided by the Pest Management Regulatory Agency
Exhibit 1.1—text version
This flow chart summarizes the regulatory process for pesticides.
Pre-market evaluation—In the pre-market evaluation of a pesticide, the Pest Management Regulatory Agency assesses the pesticide’s risks and value to determine whether they are acceptable.
Denial of registration—If the Agency deems the risks and value unacceptable, it denies the registration.
Full registration—If the Agency deems the risks and value acceptable and needs no additional data to confirm its assessment, it grants a full registration.
Re-evaluation—The Agency re-evaluates the risks and value of a pesticide every 15 years, using current scientific methods.
Conditional registration—If the Agency requires additional data from the registrant to confirm its assessment that the pesticide’s risks and value are acceptable, it grants a conditional registration.
- If the registrant does not provide the additional data within the specified time period, the Agency can either cancel or extend the conditional registration.
- If the registrant provides the additional data, the Agency reviews it for completeness and quality.
- If the Agency finds that the data is incomplete or of insufficient quality, it can either cancel or extend the pesticide’s conditional registration.
- If the Agency finds that the data is complete and of sufficient quality, it assesses the data to determine whether it confirms its initial assessment of the pesticide. If the data confirms the initial assessment, the Agency grants a full registration. If the data does not confirm the initial assessment, the Agency cancels the registration.
Special review—If new information about a registered pesticide’s risks and value is identified, the Agency may perform a targeted review to redetermine whether the pesticide’s risks and value are acceptable.
1.5 Over the past 12 years, the Commissioner of the Environment and Sustainable Development conducted two audits of the Pest Management Regulatory Agency’s management of pesticides. In the Commissioner’s 2003 Report, Chapter 1—Managing the Safety and Accessibility of Pesticides, we noted that the Agency occasionally skipped steps during its evaluations of pesticides, contrary to its prescribed process. We also noted that some required data had not been received from applicants or did not meet acceptable quality standards, and that assumptions for how pesticides were being used were not tested. Moreover, we reported that the Agency was relying heavily on temporary registrations for products that had not yet met the conditions required for full registration, and that the Agency was extending the length of these temporary registrations.
1.6 In the Commissioner’s 2008 March Status Report, Chapter 2—Chemicals Management—Pesticide Safety and Accessibility, we followed up on our 2003 audit findings, focusing on the Agency’s evaluation and re-evaluation activities. We found that the Agency had made progress in addressing deficiencies in its evaluations of new pesticides and in establishing guidelines for removing older pesticides with unacceptable risks from the market. We also found some important weaknesses. For instance, the Agency continued to make heavy use of “conditional” registrations (formerly called temporary registrations). This practice allowed registrants to place their products on the market before they had submitted the studies and data that the Agency required to confirm its risk assessments. Some conditional registrations were in place for up to 20 years. In addition, the Agency had made unsatisfactory progress in fulfilling its regulatory obligation to re-evaluate pesticides that were registered in Canada before 1995.
Focus of the audit
1.7 This audit focused on whether the Pest Management Regulatory Agency (the Agency) managed selected aspects of its mandate in accordance with the Pest Control Products Act, to prevent unacceptable risks to the health of Canadians and the environment.
Specifically, we examined whether the Agency had
- made progress in limiting the duration of conditional registrations since our 2008 audit;
- initiated and completed re-evaluations of older pesticides in a timely manner and initiated special reviews, as required by the Act;
- considered in its re-evaluations of pesticides their risks to human health and the environment, and their value, and tested the assumptions it used in its assessments;
- taken timely action to cancel the registrations of pesticides when their risks were found to be unacceptable; and
- provided the public with the required information on conditional registrations, as called for by the Pest Control Products Act, and communicated information to the public on pesticide risks.
1.8 This audit is important because these aspects of the Agency’s mandate are essential to fulfilling its primary objective of preventing unacceptable risks to people and the environment from the use of pesticides.
1.9 More details about the audit objective, scope, approach, and criteria are in About the Audit at the end of this report.
Findings, Recommendations, and Responses
Conditional registrations of pesticides
The Agency had not made progress in limiting the duration of some conditional registrations
1.10 Overall, we found that the Pest Management Regulatory Agency (the Agency) allowed conditionally registered pesticides to be in use for lengthy periods—in many cases, for more than the five-year period it normally allowed—without having received and assessed the required studies and data. As a result, the Agency could not confirm its risk or value assessments. In addition, the Agency had never exercised its authority to cancel a conditional registration when the registrant failed to satisfy the conditions.
1.11 This finding is important because while a conditionally registered product is in use, sometimes for a prolonged period, the Agency has not confirmed one or more aspects of its risk assessment. Furthermore, users may come to depend on a product that is ultimately shown to be unsafe. Market dependence and the lack of alternatives could make it more difficult for the Agency to cancel the registration of products that are later recognized to pose unacceptable risks. Eight of nine products that have been conditionally registered for more than a decade belong to the neonicotinoid class of pesticides. These products are now used extensively in Canada and are widely suspected of being a threat to bees, other pollinators, and broader ecosystems.
1.12 Our analysis supporting this finding presents what we examined and discusses
- the duration of conditional registrations, and
- the Agency’s response to unfulfilled conditions of registration.
1.13 In its initial evaluation of a pesticide, the Pest Management Regulatory Agency assesses the product’s risks to human health and the environment, and its value. The Agency may conclude that it needs additional data to confirm its assessment. In these cases, the product is granted a conditional registration. While a pesticide is conditionally registered, it can be sold and used, and conditional registration may be granted to other pesticides with the same active ingredient. Although the Pest Control Products Act places no limits on the length of time that a pesticide may be conditionally registered, the Agency normally allows the registrant three years to submit the specified information, and an additional two years for the Agency to review this information.
1.14 In our 2008 audit, we found that the duration of conditional registrations sometimes stretched to 10 or 20 years—a length of time that could not be considered temporary. The audit questioned the appropriateness of this practice, and the Agency committed to taking action to manage the use and duration of conditional registrations.
1.15 Of the approximately 7,000 pest control products registered in Canada, 80 are conditionally registered.
1.16 Our recommendation in this area of examination appears at paragraph 1.21.
1.17 What we examined. We examined whether the Agency had made progress since 2008 in reducing the length of time it allowed pesticides to remain conditionally registered.
1.18 The duration of conditional registrations. We found that although the number of pest control products with conditional registrations had decreased significantly since our 2008 audit (from 272 to 80), the duration of conditional registrations remained a concern. Since 2008, over one third of products remained conditionally registered for longer than the five-year period normally allowed by the Agency. Of the 80 products that were conditionally registered at the time of our audit, 29 had been conditionally registered for five or more years. Of these 29 products, 19 were neonicotinoid pesticides (Exhibit 1.2).
Exhibit 1.2—Length of time pest control products have been conditionally registered
| Conditionally registered products | Duration of conditional registration | ||
|---|---|---|---|
| More than 10 years | 5 to 10 years | Less than 5 years | |
| Neonicotinoids | 8 | 11 | 17 |
| Other | 1 | 9 | 34 |
| Total | 9 | 20 | 51 |
1.19 We found that nine pesticides had been conditionally registered for more than 10 years—the same number found in our 2008 audit. In one of these cases, the Agency had not completed its assessment of the information provided by the registrant in 2010. In the remaining eight cases, all of which involved neonicotinoid pesticides, the Agency took 3 to 4 years to complete its assessments of the data submitted by registrants, and the assessments revealed that additional information was needed (Exhibit 1.3).
Exhibit 1.3—The Pest Management Regulatory Agency has allowed the use of neonicotinoids since 1995 without having confirmed whether the risks are acceptable

Honeybees collecting pollen
Photo: Sherjaca/Shutterstock.com
Neonicotinoid insecticides are widely used around the world. When applied to seeds before planting, they render the plant toxic to insects. These pesticides have been used in Canada since 1995 on corn, canola, soybeans, and other crops.
Growing market reliance. Currently, 36 of Canada’s 80 conditionally registered pest control products are neonicotinoids. Between 2008 and 2015, the number of conditionally registered neonicotinoid pest control products increased by 80 percent (from 20 products to 36). The growth of these products in number and sales suggests that the market has become increasingly reliant on them.
Potential risks. Neonicotinoids are now widely suspected of causing significant ecological harm. Much of the concern has focused on their potential negative effects on bees. Recent studies indicate that neonicotinoids have broader effects on ecosystems—involving, for example, birds, earthworms, other pollinators, and aquatic invertebrates.
Value questioned. The US Environmental Protection Agency recently concluded that these seed treatments provide negligible overall benefits to soybean production in most situations. In Canada, a draft study by the Pest Management Regulatory Agency (the Agency) indicated that the lost yield from a ban on neonicotinoids would have a relatively small economic cost in proportion to the overall value of corn and soybean crops.
The Agency’s actions. In 2003, the Pest Management Regulatory Agency determined that studies of the chronic toxicity of clothianidin (a neonicotinoid) to honeybee hives were required by 2008 as a condition of registration. The Agency later identified the absence of these studies as a “critical data gap.” More than a decade after granting the conditional registration, the Agency had still not confirmed its risk assessment. Despite this, the Agency continued to grant conditional registrations for clothianidin and other neonicotinoid products.
In June 2012, the Agency initiated re-evaluations of agricultural uses of neonicotinoid pesticides in collaboration with the US Environmental Protection Agency. The re-evaluations are expected to be completed in 2017 or 2018.
Mitigation measures. In the interim, the Pest Management Regulatory Agency implemented several measures for the corn and soybean planting seasons in 2014 and 2015, including the use of dust-reducing seed planting practices and new product labels with enhanced warning statements.
1.20 The Agency’s response to unfulfilled conditions of registration. We also found that the Agency had never exercised its authority to cancel a conditional registration when registrants failed to fulfill the conditions of registration. There appeared to be no consequences for registrants that did not meet the conditions.
1.21 Recommendation. The Pest Management Regulatory Agency should ensure that registrants respect the timelines specified in conditional registrations for providing required information. It should also assess the data within its established two-year timeline to determine the continued acceptability of pest control products and active ingredients.
The Agency’s response. Agreed. Since 2008, the Pest Management Regulatory Agency has taken action to limit the number of conditional registrations, which are used only where risks have been deemed acceptable and where additional, confirmatory information has been considered desirable. Currently, one percent of pesticide registrations are conditional in nature; most of these have been conditional for less than five years. Some pesticides have been conditionally registered for longer periods, mainly because the Agency has required information that would take multiple years to generate (for example, new types of scientific studies to be carried out).
The Agency believes it could be possible to eliminate the use of conditional registrations and is developing options towards that goal. Stakeholders will be consulted on any proposed change in policy.
For the remaining conditional registrations, on a biannual basis, the Agency will post a status report on conditional registrations, including whether the requested information has been received, the actions that have been taken, and the performance in reviewing the data within the two-year timeline. Where necessary, the Agency will use existing authorities in the Pest Control Products Act to ensure that the requested information is provided according to the established timelines.
Re-evaluations of pesticides
1.22 Overall, we found that the Pest Management Regulatory Agency (the Agency) did not manage an important aspect of its re-evaluations of pesticides according to the Pest Control Products Act. Although the Agency considered the value of pesticides and their health and environmental risks, it did not assess their cumulative effects on human health when required by the Act. In addition, the Agency had made insufficient progress in completing the re-evaluations of older pesticides.
1.23 These findings are important because the Agency must apply the latest available scientific knowledge and data to its re-evaluations to confirm that the health and environmental risks of older pesticides remain within acceptable limits. If the Agency does not consider the cumulative health effects of pesticides in its re-evaluations, it lacks potentially important information about risks and how they should be managed. The Agency’s slowness in completing risk assessments may cause product labels and mitigation measures to become outdated, and pesticides remaining on the market may no longer meet current standards. Such situations could expose users and the environment to unacceptable risks.
1.24 Pest control products must be periodically re-evaluated to ensure that risk assessments and mitigation or precautionary measures reflect the latest scientific knowledge. The Pest Control Products Act requires the Pest Management Regulatory Agency to re-evaluate pesticides that were registered before 1995 and to re-evaluate all pesticides on a 15-year cycle. These requirements are aimed at ensuring that the instructions on product labels are updated as necessary to protect human health and the environment, and that pesticides posing unacceptable risks are removed from the Canadian market.
Risk to human health—The possibility of harm to human health resulting from exposure to or use of the product, taking into account its conditions or proposed conditions of registration.
Source: Pest Control Products Act
Risk to the environment—The possibility of harm to the environment, including its biological diversity, resulting from exposure to or use of the product, taking into account its conditions or proposed conditions of registration.
Source: Pest Control Products Act
Value—The product’s actual or potential contribution to pest management, taking into account its conditions or proposed conditions of registration, and includes the product’s (a) efficacy; (b) effect on host organisms in connection with which it is intended to be used; and (c) health, safety, and environmental benefits and social and economic impact.
Source: Pest Control Products Act
1.25 Pesticides are re-evaluated to determine whether and under what conditions their continued registration is acceptable. The active ingredient and the associated pest control products are considered in terms of their risks to human health, risks to the environment, and value.
1.26 Since 2006, the Pest Control Products Act has required the Agency to take into account aggregate exposure and cumulative health effects, and to apply margins of safety, when it re-evaluates health risks:
- Aggregate exposure is the overall exposure to a single pesticide that can occur from food, drinking water, residential and other non-occupational sources, and all known or plausible routes of exposure (that is, mouth, skin, and inhalation).
- Cumulative health effects are caused by exposure from pest control products that have a common mechanism of toxicity and thereby result in a common toxic effect (for example, products that affect the functioning of an enzyme in the nervous system).
- Margins of safety are applied to take into account the use of data from animal studies and the differences between human and animal populations, in addition to the sensitivities of major identifiable subgroups, such as women, pregnant women, infants, children, and seniors.
1.27 To determine whether a pesticide has acceptable risks, the Agency’s evaluators must make a series of assumptions to link laboratory studies to the potential impacts of the pesticide on human health and the environment. These assumptions then must be tested to validate the Agency’s risk assessments. Monitoring the exposure and effects of pesticides is an important aspect of testing assumptions.
1.28 In 2003, we reported that key assumptions were not tested, and that some assumptions were not correct. In 2008, we reported that the Agency was making progress in testing its assumptions.
In its re-evaluations, the Agency considered the risks and value of pesticides, but had not assessed their cumulative health effects when required
1.29 We found that the Pest Management Regulatory Agency considered the value of pesticides, and their risks to human health and the environment, in its re-evaluations. However, the Agency had not assessed the cumulative health effects of pesticides when required. In addition, we found that the Agency worked with other federal departments, international partners, the pesticide industry, and academia to validate the assumptions it used in its risk assessments.
1.30 Our analysis supporting this finding presents what we examined and discusses
- risks and value,
- aggregate exposure,
- cumulative health effects,
- margins of safety, and
- assumption testing.
1.31 This finding matters because a lack of information about the cumulative health effects of pesticides could affect the Pest Management Regulatory Agency’s decisions on whether risks are acceptable and how risks should be managed.
1.32 Our recommendation in this area of examination appears at paragraph 1.47.
1.33 What we examined. We examined whether the Pest Management Regulatory Agency, in its re-evaluations, considered the value of pesticides and their health and environmental risks, as required by the Pest Control Products Act. We also examined the extent to which the Agency’s re-evaluations of health risks complied with the new aspects of the Act—that is, whether the Agency took into account aggregate exposure and cumulative health effects, and applied margins of safety. Finally, we examined whether the Agency had ensured that the assumptions it used in its risk and value assessments were valid.
1.34 Risks and value. The Pest Control Products Act requires the Agency to re-evaluate active ingredients in pest control products on the basis of updated information and current standards to determine whether and under what conditions continued registration is warranted.
1.35 We examined 10 re-evaluations of active ingredients that were completed by the Agency in the 2014–15 fiscal year to determine whether the Agency considered health and environmental risks, and value, in its assessments.
1.36 All 10 of these re-evaluations resulted in changes to the instructions on product labels to mitigate risk and ensure the application of current standards and practices. For example, the revised instructions specified the need for buffer zones between fields and streams, or additional personal protective gear. In 2 of these re-evaluations, the active ingredients were found to pose unacceptable risks to human health, and certain uses of the products were cancelled as a result.

Use of personal protective gear to mitigate risks to pesticide user
Photo: wellphoto/Shutterstock.com
1.37 We found that the Agency considered risks and value in the 10 re-evaluations we examined. In 3 of these re-evaluations, the Agency considered new information from the registrant as well as published literature. In the remaining 7 re-evaluations, the Agency decided that its existing assessments were sufficient because the earlier assessments met current scientific standards, or because the active ingredient had a limited use pattern.
1.38 Aggregate exposure. According to the Pest Control Products Act, human health risk assessments of pesticides must include consideration of the combined or “aggregate” exposure from dietary, residential, and drinking water sources.
1.39 We found that the Pest Management Regulatory Agency had considered the need to conduct an aggregate exposure assessment in the 10 re-evaluations we examined. In 4 re-evaluations, the Agency conducted an assessment of aggregate exposure. In the remaining 6 re-evaluations, the Agency determined that this assessment was not required. For example, the assessment would not be required for a pesticide that was not registered for food or residential uses, because it would not have multiple routes of exposure to be assessed.
1.40 Cumulative health effects. The Pest Control Products Act requires the assessment of cumulative health effects when two or more pesticides have been proven to share a common mechanism of toxicity.
1.41 The Agency concluded that a cumulative risk assessment was not warranted in 6 of the 10 re-evaluations we examined. However, in some of the 6 cases, we found no evidence to support the Agency’s conclusion that there was no common mechanism of toxicity. For 2 of the other 4 re-evaluations, we found that the Agency had yet to determine whether a cumulative risk assessment was warranted. For the 2 remaining re-evaluations, the Agency had determined that the assessment was warranted, but it had not completed the work.
1.42 Since 2006, the Pest Control Products Act has required the Agency to take cumulative health effects of pesticides into account when it re-evaluates risks. The Agency informed us that there was no global consensus on how to assess cumulative exposure, and that information was typically not available to determine which chemicals act in the same manner. In 2012, the Agency began work to develop a methodology for cumulative assessment, scheduled for completion in the 2017–18 fiscal year.
1.43 Margins of safety. We found that in the 10 re-evaluations we examined, the Pest Management Regulatory Agency applied margins of safety according to its policy. This practice takes into account the use of data from animal studies and the differences between human and animal populations, in addition to the sensitivities of major identifiable subgroups, such as women, pregnant women, infants, children, and seniors.
1.44 The Agency also considered whether to apply an additional protection factor for effects on infants and children, as required by the Pest Control Products Act. In 1 of the 10 re-evaluations we examined, the Agency applied this additional protection factor. In the other 9 re-evaluations, the Agency concluded that it had the data it needed to determine that this additional protection factor was either not applicable or not required.
1.45 Assumption testing. The Pest Management Regulatory Agency relies on other federal departments, international partners, the pesticide industry, and academia to generate the research and monitoring information it needs to verify assumptions. We found that since 2008, the Agency worked with these groups to validate the assumptions it used in its risk assessments. Some of this work included research on environmental effects of specific pesticides, including the effects of the herbicide glyphosate on amphibians, and on the effectiveness of mitigation measures, such as buffer zones to reduce runoff into aquatic areas.
1.46 The Agency also had additional work under way to verify and refine assumptions. Its priorities included monitoring the pesticide residue on food and pesticide concentrations in water, and researching the effects of pesticides used in aquaculture.
1.47 Recommendation. The Pest Management Regulatory Agency should finalize and apply the methodology required to ensure that it considers cumulative health effects of pesticides in its re-evaluations, as required by the Pest Control Products Act.
The Agency’s response. Agreed. The Pest Management Regulatory Agency agrees with the recommendation and the importance of ensuring that the cumulative effects of pesticides are considered where necessary.
This is an emerging area in the science used to regulate pesticides, and the Agency is actively working with international partners to understand how to best apply these principles to protect the health and safety of Canadians. Most countries, including Canada, are working on a cumulative risk assessment methodology for pesticides. However, the international scientific community has not yet reached a consensus on how to best assess the cumulative effects of pesticides.
The Agency anticipates having its own methodology for cumulative risk assessments in place in the 2017–18 fiscal year.
The Agency made insufficient progress in completing re-evaluations of older pesticides
1.48 We found that the Pest Management Regulatory Agency remained slow in completing the re-evaluations of pesticides that were registered before 1995. We are concerned that the Agency will not be able to reduce its workload of re-evaluations unless it accelerates its pace of work.
1.49 Our analysis supporting this finding presents what we examined and discusses
1.50 This finding matters because the Agency conducts re-evaluations to ensure that the human health and environmental risks of pesticides remain within acceptable limits according to the latest available scientific knowledge and data. For 95 percent of the re-evaluations carried out to date, the Agency decided that additional precautions were necessary to protect human health and the environment. When the Agency is slow to complete its re-evaluations, its risk assessments, labels, and mitigation measures may become outdated, potentially posing unacceptable risks to users and the environment.
1.51 Our recommendations in this area of examination appear at paragraphs 1.61 and 1.64.
1.52 What we examined. We examined the Pest Management Regulatory Agency’s progress in completing re-evaluations of older pesticides since our 2008 audit, and in developing management tools for planning and tracking the status of its re-evaluations.
1.53 Completing re-evaluations of older pesticides. Many active ingredients that have been registered in Canada for decades have not been re-evaluated according to today’s more stringent methods and standards.
1.54 In 2001, the Agency committed to re-evaluating 401 active ingredients that were registered before 1995. Ultimately, 287 active ingredients needed to be re-evaluated, because many of the 401 active ingredients had been discontinued or were no longer used in pest control products.
1.55 The Agency initially set a 2006 completion date for these re-evaluations. Since 2001, the Agency changed this date twice: first to the 2008–09 fiscal year, and then to the 2009–10 fiscal year.
1.56 We found that the Agency had completed 241 of the 287 re-evaluations. Although the Agency had taken about 4 years, on average, to complete a re-evaluation, we found that the 46 re-evaluations not yet completed (16 percent) had been under way for about 10 years, including the re-evaluation of chlorpyrifos (Exhibit 1.4). When we began our audit, we found that the Agency had not set target completion dates for half of these re-evaluations. By the end of our audit period, it had set target completion dates for all of these re-evaluations.
Exhibit 1.4—The re-evaluation of chlorpyrifos was initiated in 1999 but remains incomplete
Chlorpyrifos was first registered in Canada in 1969 to control pests on many food crops, including wheat and potatoes.
In 1999, the Pest Management Regulatory Agency (the Agency) initiated a re-evaluation of chlorpyrifos. By 2002, the Agency had discontinued many residential uses of pesticides that contained this active ingredient, as a way to mitigate risks. In the following year, the Agency consulted with the public on its proposed decision on the remaining uses. However, more than a decade later, the re-evaluation of chlorpyrifos remains incomplete, and no final decisions have been made about the remaining uses.
In its re-evaluation, the Agency found that chlorpyrifos is acutely toxic to a wide range of organisms, including mammals, birds, fish, aquatic invertebrates, and honeybees. In 2003, as a result of these risks to the environment, the Agency proposed to cancel the use of chlorpyrifos on a number of crops, including corn, filberts, lentils, oats, peppers, sugar beets, tobacco, peaches, and nectarines.
In 2007, after public consultation, the Agency decided to reverse the cancellation for all of these crops, citing a lack of alternative pest control products.
The Agency also planned to complete a refined environmental assessment by 2008. This assessment is still outstanding. The Agency plans to publish its decision in 2020.
1.57 The Agency published interim mitigation measures for 17 of the 46 incomplete re-evaluations. These interim measures were intended to reduce risks to human health and the environment while the Agency finalized its assessments.
1.58 An additional 52 re-evaluations of active ingredients registered in 1995 or later were to be initiated between 2011 and 2015, as required by the Pest Control Products Act. When we began our audit, we found that 12 of these re-evaluations had not yet been initiated. By the end of our audit period, all 12 re-evaluations had been initiated.
1.59 We also found that the Agency had not cleared its backlog of incomplete re-evaluations of pesticides registered before 1995 (Exhibit 1.5). Over the past five fiscal years, the Agency completed about 14 re-evaluations per year. As of October 2015, 87 re-evaluations were under way. At that rate, the Agency had approximately six years of work in progress, and it planned to initiate an additional 43 re-evaluations in the next two fiscal years. We are concerned that if the Agency does not accelerate its pace of work, it will be unable to reduce the number of incomplete re-evaluations.
Exhibit 1.5—The Pest Management Regulatory Agency has not kept pace with its workload of re-evaluations
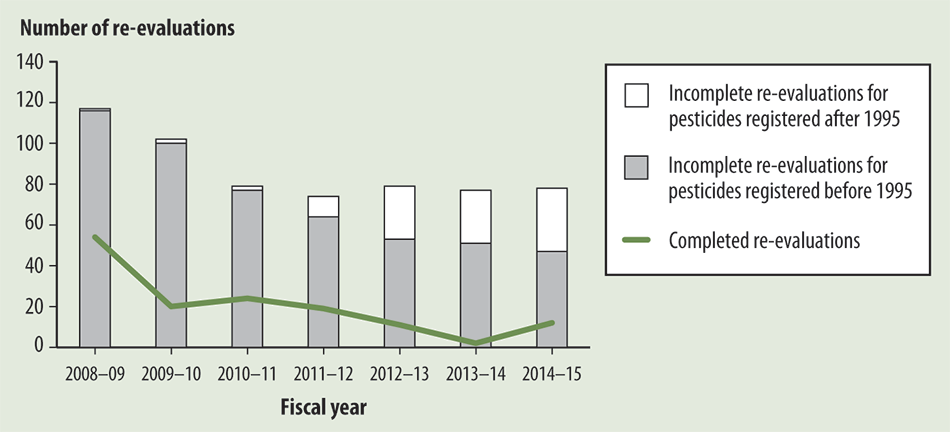
Source: Based on data provided by the Pest Management Regulatory Agency
Exhibit 1.5—text version
This bar graph shows the Pest Management Regulatory Agency’s re-evaluation workload for the past seven fiscal years. The number of re-evaluations for each fiscal year is divided into three groups:
- incomplete re-evaluations for pesticides registered after 1995,
- incomplete re-evaluations for pesticides registered before 1995, and
- completed re-evaluations.
Since the 2008–09 fiscal year, the number of incomplete re-evaluations for pesticides registered after 1995 has increased, whereas the number of incomplete re-evaluations for pesticides registered before 1995 has decreased. The number of completed re-evaluations has decreased overall since the 2008–09 fiscal year, with slight increases occurring between the 2009–10 and 2010–11 fiscal years and the 2013–14 and 2014–15 fiscal years.
| Fiscal year | Incomplete re-evaluations for pesticides registered before 1995 | Incomplete re-evaluations for pesticides registered after 1995 | Completed re-evaluations |
|---|---|---|---|
| 2008–09 | 116 | 1 | 54 |
| 2009–10 | 100 | 2 | 20 |
| 2010–11 | 77 | 2 | 24 |
| 2011–12 | 64 | 10 | 19 |
| 2012–13 | 53 | 26 | 11 |
| 2013–14 | 51 | 26 | 2 |
| 2014–15 | 47 | 31 | 12 |
1.60 Moreover, nearly all of the completed re-evaluations (95 percent) resulted in label changes to mitigate risk. These changes included revisions to the instructions for use and restrictions for some uses. It seems likely, therefore, that the pesticides that have yet to be re-evaluated pose health or environmental risks that will need to be addressed by adding precautionary measures or by cancelling their registrations for some or all uses.
1.61 Recommendation. The Pest Management Regulatory Agency should establish realistic timelines for completing its re-evaluations of active ingredients registered before 1995, and should complete them accordingly.
The Agency’s response. Agreed. The Pest Management Regulatory Agency has completed the majority of its re-evaluations of active ingredients registered before 1995 and is committed to completing the remainder of the re-evaluations by 2020. The Agency continues to make progress on the remaining 46 and, where necessary, has implemented interim measures to further protect health and the environment during the review process.
The Minister of Health’s multi-stakeholder Pest Management Advisory Council recently supported the Agency’s three-year re-evaluation work plan. The Agency will update the current work plan, in collaboration with the Council, in order to ensure that the remaining 46 re-evaluations are completed by 2020. In completing the re-evaluations, the Agency will collaborate with other foreign regulators, as appropriate, including the US Environmental Protection Agency.
The Pest Management Regulatory Agency will continue to report on progress on an annual basis.
1.62 Planning and tracking re-evaluations. We found that the Pest Management Regulatory Agency’s approach to planning and tracking the status of re-evaluations was not systematic. Information identifying which re-evaluations needed to be done, and when, was missing from the Agency’s schedule of work and other documents the Agency used to track the status of its re-evaluations.
1.63 However, once the Agency had initiated a re-evaluation of a pesticide registered after 1995, it published individual project plans, which included timelines for completing the necessary work. The Agency’s standard timeline ranged from about 1 to 4 years, depending on the level of complexity.
1.64 Recommendation. With respect to its overall work planning for re-evaluations, the Pest Management Regulatory Agency should ensure that its schedule of work is complete and up to date.
The Agency’s response. Agreed. The Pest Management Regulatory Agency will publish updated work plans in early 2016 and update them on a yearly basis. These will include clear expectations for timelines, as well as the expected publication date of the final re-evaluation decision.
The Agency’s current approach to re-evaluation favours receipt of new information, including incident reports, from any and all stakeholders even where this material might arrive very late in the review process. While this approach helps ensure re-evaluations are based on the latest and most complete set of information available, it unfortunately often delays the final decision, as the Agency must then reconsider its assessment. To address this challenge and to meet pre-established timelines, the Agency will consult on and develop a policy with clear rules and criteria for submitting information in order to make timely decisions.
Special reviews of pesticides
The Agency had begun to consider whether special reviews were warranted for pesticides banned since 2013 in OECD countries
1.65 Overall, we found that the Pest Management Regulatory Agency (the Agency) was in the process of deciding whether special reviews of pesticides banned for all uses between June 2013 and December 2014 in other Organisation for Economic Co-operation and Development (OECD) countries were warranted.
1.66 This finding is important because special reviews are intended to enable the Agency to act promptly on new information about risks to human health and the environment that may emerge in the 15-year period between initial registration and re-evaluation. If special reviews are not conducted in a timely manner, products with unacceptable risks may remain in use in Canada, or be used without appropriate mitigation or precautionary measures.
1.67 Our analysis supporting this finding presents what we examined and discusses
1.68 A special review allows the Pest Management Regulatory Agency to consider new information on emerging risks at any time during the registration of a pesticide. The Pest Control Products Act requires the Agency to conduct a special review when an OECD member country prohibits all uses of an active ingredient for health or environmental reasons. The Agency must also initiate a special review of a registered pesticide if the Minister of Health has reasonable grounds to believe that the pesticide has unacceptable risks or value. According to the Act, a member of the public may request a special review. The Agency may also initiate a special review on the basis of information provided by a federal or provincial government department or agency.
1.69 We made no recommendations in this area of examination.
1.70 What we examined. We examined the Pest Management Regulatory Agency’s work in initiating special reviews.
1.71 Initiation of special reviews. In 2006, the Agency received a request to initiate 60 special reviews of active ingredients that were banned in other OECD countries. A year later, the Agency responded that it did not need to initiate the special reviews, because the registrations of these active ingredients had been cancelled, had recently been re-evaluated, or were undergoing a re-evaluation. In 2011, the Federal Court concluded that re-evaluations could not replace special reviews of pesticides.
1.72 Following court proceedings in 2013, the Agency initiated 23 special reviews of active ingredients that had been banned for all uses in other OECD countries. The 2006 request included 15 of these special reviews. The Agency had also initiated 4 additional special reviews as a result of public requests.
1.73 In 2014, the Agency developed a standard operating procedure with timelines for determining whether to conduct special reviews—eight years after provisions for special reviews were included in the Pest Control Products Act. We found that, because the procedure had only recently been developed, there were delays in the Agency’s determining whether to initiate additional special reviews of pesticides banned for all uses in other OECD countries. The Agency informed us that it was reviewing information on three OECD-banned pesticides that it had received between June 2013 and December 2014 to determine whether special reviews were warranted. It was too early to tell whether the Agency was respecting the timelines set out in its new procedure.
Cancellation of pesticide registrations
The Agency did not promptly cancel the registrations of some pesticides whose risks it had deemed unacceptable
1.74 Overall, we found that the Pest Management Regulatory Agency (the Agency) did not promptly cancel the registrations of some pesticides when it determined that they posed unacceptable risks. In all but one case, the Agency took between 4 and 11 years to cancel the registrations. In several cases, the Agency cited the lack of alternatives as the reason for delaying cancellations. In other cases, the cancellations were delayed to allow suppliers and users to exhaust their inventories.
1.75 Our findings are important because lengthy delays in cancelling the registration of pesticides that have been found to pose unacceptable risks that cannot be mitigated prolong the time that workers, the public, and the environment are exposed to these risks. The Pest Control Products Act states that the Agency’s primary objective is to prevent unacceptable risks to people and the environment from the use of pest control products. Lengthy delays are contrary to the Agency’s statutory objective.
1.76 Our analysis supporting this finding presents what we examined and discusses
1.77 The Pest Management Regulatory Agency may decide, as a result of its re-evaluations or special reviews, that a pesticide’s active ingredient poses unacceptable health or environmental risks that cannot be mitigated. This means that a pesticide with this active ingredient would never have been registered if assessed according to current scientific knowledge.
Cancellation date—The date on which pest control products containing a specific active ingredient can no longer be sold or used.
1.78 According to the Pest Control Products Act, after the Agency determines that an active ingredient poses an unacceptable risk, it may immediately cancel the registration, or it may set a delayed cancellation date.
1.79 Our recommendation in this area of examination appears at paragraph 1.84.
1.80 What we examined. We examined the Pest Management Regulatory Agency’s activities since 2008 that were associated with the cancellation of registrations of seven active ingredients. After applying current scientific knowledge in its re-evaluations of these active ingredients, the Agency had determined that they posed risks that were unacceptable for all uses and could not be mitigated.
1.81 Time frames for cancelling registrations. The Agency identified several factors it considered when establishing cancellation dates. These included the availability of alternatives, the time allowed for suppliers and users to exhaust their inventories, and the nature and severity of human health or environmental risks.
1.82 We found that the Agency did not always act promptly to cancel the registrations of the active ingredients or associated products that we examined. Although the Agency set schedules for these pesticides to be phased out over a period of years, it often extended the phase-out periods (Exhibits 1.6 and 1.7).
Exhibit 1.6—The Pest Management Regulatory Agency took 11 years to cancel the registration of a pesticide after finding its risks unacceptable

A damaged potato from late summer wireworm feeding (left) compared with a healthy one (right).
Photo: R.S. Vernon
Phorate is an active ingredient that was registered in Canada in 1962. The Pest Management Regulatory Agency (the Agency) initiated a re-evaluation of phorate in 1999 and completed it in 2004. The re-evaluation concluded that this insecticide had high acute toxicity and posed risks that made it unacceptable for all uses. Its effects on the environment could not be mitigated, because even small doses could be harmful.
The Agency phased out the use of phorate on some crops, including corn and lettuce, in 2004. It also decided to phase out the use of this pesticide on potatoes by 2006. However, it extended this time frame four times: to 2007, 2008, 2012, and then 2015. The Agency cited the lack of alternative methods to control wireworm on potatoes as the reason for the extensions.
In August 2015, more than a decade after the Agency had determined that this pest control product posed unacceptable risks, the Agency cancelled the registration. During the audit, the Agency informed us that it had registered an alternative formulation of phorate that mitigated the risks.
Exhibit 1.7—The Pest Management Regulatory Agency allowed lengthy phase-out periods for some pesticides
| Active ingredient (year registered) |
Year of decision to cancel registration | Year of cancellation | Phase-out period (years) |
|---|---|---|---|
| Tributyltin oxide (1955) | 2010 | 2014 | 4 |
| Azinphos-methyl (1958) | 2004 | 2012 | 8 |
| Endosulfan (1958) | 2011 | 2016a | 5a |
| Phorate (1962) | 2004 | 2015b | 11 |
| Carbofuran (1969) | 2010 | 2012 | 2 |
| Terbufos (1979) | 2004 | 2012 | 8 |
| Tributyltin maleate (1979) | 2010 | 2014 | 4 |
Source: Based on information provided by the Pest Management Regulatory Agency
1.83 During the audit period, the registration of one of the seven active ingredients we examined was still in the process of being cancelled, and it could still be used legally. In all but one case, the Agency took between 4 and 11 years to cancel the registrations. In several cases, the Agency cited lack of alternatives as the reason for delaying the cancellations. In other cases, the cancellations were delayed to allow suppliers and users to exhaust their inventories.
1.84 Recommendation. When the Pest Management Regulatory Agency determines that the risks posed by a pesticide are unacceptable for all uses, it should act promptly to cancel the pesticide’s registration.
The Agency’s response. Agreed. The Pest Management Regulatory Agency agrees that unacceptable risks presented by pesticides should be addressed promptly.
Where unacceptable risks are identified but can be appropriately addressed through mitigation measures, the Agency acts promptly to require such measures. These can range from cancellation of certain uses, to the enhancement of the use of personal protective equipment by workers, to the implementation of additional application restrictions.
Where risks are imminent and no appropriate mitigation measures exist, the Agency’s policy is to cancel all uses.
In some circumstances where risks are only considered unacceptable in the context of very long-term usage, and where mitigation measures can be put in place as an effective interim solution, the Agency may implement a phase-out period commensurate with the level of risk. This approach ensures the protection of human health and the environment and provides lead time for improved products to be introduced. The use of orderly product phase-outs, where risks can be mitigated in the interim, is a widely accepted practice in use by the US Environmental Protection Agency and other advanced economies.
In 2016, to improve transparency, the Agency will consult publicly on its current approach for establishing a phase-out of products (including extensions).
Communication with the public
The Agency did not provide the public with complete information on conditional registrations, as required by the Pest Control Products Act, or effectively communicate new information on pesticide risks
1.85 Overall, we found that the Pest Management Regulatory Agency (the Agency) failed to maintain a complete public registry of conditionally registered products and their associated conditions. The Agency’s public registry did not make clear whether a pesticide was conditionally registered, and information on conditions of registration was sometimes missing or out of date. Other information that was not always readily available included which products the Agency had decided to remove from the market, which uses it had decided to prohibit, what mitigation measures it had decided to put in place, and when changes would take effect.
1.86 Our findings are important because Canadians need up-to-date information to make informed decisions about pesticide use. If the Agency does not provide required information and communicate risks effectively, Canadians cannot make informed decisions on how to use products safely.
1.87 Our analysis supporting this finding presents what we examined and discusses
1.88 The Pest Management Regulatory Agency’s key objectives are to protect human health and the environment, and to support Canadian competitiveness, by regulating pesticides and their use effectively and transparently.
1.89 The Pest Control Products Act requires the Agency to provide the public with information about pest control products and their conditions of registration through an electronic registry.
1.90 Our recommendations in this area of examination appear at paragraphs 1.93 and 1.97.
1.91 What we examined. We examined the availability of information on conditional registrations, re-evaluations, and cancellations of registrations.
1.92 Identifying conditional registrations and their status. We found that the Agency did not inform the public about which products were conditionally registered or what their associated conditions of registration were, as required by the Pest Control Products Act. Information on the data or studies that the Agency required from the registrants was missing for 25 of the 80 conditionally registered products. Information was provided for the remaining 55 conditionally registered products. However, for 22 of these 55 products, the deadlines for submitting the required data or studies had expired, and no update on these products was provided, leaving users with no information on their registration status. As a result, the public was not given a complete picture of the status of pest control products with conditional registrations.
1.93 Recommendation. The Pest Management Regulatory Agency should maintain its public registry of pest control products, as required by the Pest Control Products Act, to clearly communicate which products are conditionally registered and what their associated conditions of registration are.
The Agency’s response. Agreed. At present, while some information on which products are conditionally registered and their associated conditions is currently available on the Pest Management Regulatory Agency’s section of Health Canada’s website, other information is provided to members of the public upon request.
Recognizing the importance of promoting greater transparency in the regulation of pesticides, the Pest Management Regulatory Agency is constantly striving to find ways to more effectively communicate information on conditional registrations with regulated parties, affected stakeholders, and all Canadians. In 2016, all conditions associated with conditional registrations will be published on the Health Canada website.
1.94 Communicating re-evaluation decisions. We found that, although the Agency posted its re-evaluation decisions for active ingredients on Health Canada’s website and through its Really Simple Syndication (RSS) feed, other information was not always readily accessible to the public—for instance, which products were to be removed from the market, or when these changes would take effect.
1.95 We found that delays occurred—sometimes for up to two years—from the time the Agency made its re-evaluation decisions to the time it required product labels to be updated to reflect required changes, such as the need for new protective equipment or the planned phase-outs of some uses. Additional delays might occur because retailers were allowed to sell the associated products with the outdated labels, to exhaust their inventories. As a result of these delays in making label changes and the lack of readily accessible information on re-evaluation decisions, Canadians did not have timely information on how to use the pest control products appropriately.
1.96 One case in which information was not readily available to the public involved propoxur—an active ingredient that controls a range of insects, including cockroaches, ants, fleas, and ticks. In 2014, after re-evaluating this active ingredient, the Agency decided to prohibit several of its uses, including its use in pet collars. We found that the Agency’s decision did not identify which specific products would be removed from the market or when they would be removed. In addition, the label information on these products had not been updated to reflect the Agency’s decision.
1.97 Recommendation. To ensure that end users can make informed decisions on pesticide use, the Pest Management Regulatory Agency should consistently communicate timely and readily accessible information on
- which products it has decided to remove from the market,
- which uses it has decided to prohibit,
- what mitigation measures it has decided to put in place, and
- when these changes will take effect.
The Agency’s response. Agreed. The Pest Management Regulatory Agency acknowledges that the current communication of the information can be improved by clearly identifying which products (rather than active ingredient) are implicated. Recently, the Agency has increased the clarity of its regulatory documents by including timelines for the implementation of regulatory decisions.
In 2016, the Agency will identify further opportunities to improve communication with stakeholders and the Canadian public regarding the removal of pesticides or their uses, and the associated timelines. The Agency will also revise decision documents to provide a clear summary of the products it has decided to remove from the market, what uses it has decided to cancel, what mitigation measures it has decided to put in place, and when these changes will take effect.
Conclusion
1.98 We concluded that the Pest Management Regulatory Agency considered health and environmental risks, and value, in re-evaluating pesticides. However, with respect to conditional registrations, re-evaluations, special reviews, and cancellations of registrations, we concluded that the Agency had not always acted in a timely manner to fulfill its statutory objective of preventing unacceptable risks to the health of Canadians and the environment from the use of pesticides.
About the Audit
The Office of the Auditor General’s responsibility was to conduct an independent examination of selected aspects of the Pest Management Regulatory Agency’s (the Agency’s) management of pesticides, to provide objective information, advice, and assurance to assist Parliament in its scrutiny of the government’s management of resources and programs.
All of the audit work in this report was conducted in accordance with the standards for assurance engagements set out by the Chartered Professional Accountants of Canada (CPA) in the CPA Canada Handbook—Assurance. While the Office adopts these standards as the minimum requirement for our audits, we also draw upon the standards and practices of other disciplines.
As part of our regular audit process, we obtained management’s confirmation that the findings in this report are factually based.
Objective
The audit objective was to determine whether the Pest Management Regulatory Agency was managing conditional registrations, re-evaluations, special reviews, and the removal of pesticides from the market in accordance with the Pest Control Products Act, to prevent unacceptable risks to the health of Canadians and the environment.
Scope and approach
The audit focused on Health Canada’s Pest Management Regulatory Agency, which is responsible for regulating pesticides in Canada. Our audit included interviews with Agency staff, and with key stakeholders in the area of pesticides. In addition, we reviewed and analyzed documentation provided by the Agency.
To assess whether the Agency had reduced the length of time it allowed for products to remain conditionally registered, we compared the duration of conditional registrations during our audit period to the duration of those from our 2008 audit period.
To assess whether the Agency had considered risks to human health and the environment, and value, in its re-evaluations of pesticides, we examined the 10 re-evaluations of active ingredients that the Agency completed in the 2014–15 fiscal year. We performed a file review and looked at key policies and procedures related to re-evaluations. We also looked at the Agency’s progress in completing re-evaluations of pesticides registered before 1995 and of pesticides registered since then.
To assess whether the Agency was promptly cancelling the registration of unacceptable pesticides, we examined its activities since 2008 that were associated with the cancellation of registrations of seven active ingredients.
Criteria
To determine whether the Pest Management Regulatory Agency (the Agency) was managing conditional registrations, re-evaluations, special reviews, and the removal of pesticides from the market in accordance with the Pest Control Products Act (the Act), to prevent unacceptable risks to the health of Canadians and the environment, we used the following criteria:
| Criteria | Sources |
|---|---|
|
The Agency has made progress since 2008 in the use of conditional registrations by reducing the duration of conditionally registered pesticides. |
|
|
The Agency receives the additional information it requires of the registrant within the specified time frame (in the Section 12 notices). |
|
|
The Agency has met its target to re-evaluate pesticides that were approved before 1 April 1995. |
|
|
The Agency re-evaluates pesticides approved after 1995 on a 15-year cycle according to its established milestone targets and timelines. |
|
|
The Agency applies its directive for carrying out special reviews. |
|
|
In its re-evaluations, the Agency considers the health and environmental risks, and the value, of pest control products. |
|
|
In its re-evaluations, the Agency ensures that assumptions it uses in risk and value assessments are realistic and tested. |
|
|
The Agency takes action in accordance with the Act to ensure that pest control products that it has determined should no longer be registered for use in Canada are removed from the market. |
|
Management reviewed and accepted the suitability of the criteria used in the audit.
Period covered by the audit
The audit covered the seven-year period from our last audit in March 2008. Elements of the audit focused on the most recent fiscal year, 2014–15, to ensure that we examined the Pest Management Regulatory Agency’s current practices. Audit work for this report was completed on 27 October 2015.
Audit team
Principal: Andrew Ferguson
Director: Doreen Deveen
Amélie Beaupré-Moreau
Kristin Lutes
Marie-Soleil Nappert
Erin Windatt
List of Recommendations
The following is a list of recommendations found in this report. The number in front of the recommendation indicates the paragraph where it appears in the report. The numbers in parentheses indicate the paragraphs where the topic is discussed.
Conditional registrations of pesticides
| Recommendation | Response |
|---|---|
|
1.21 The Pest Management Regulatory Agency should ensure that registrants respect the timelines specified in conditional registrations for providing required information. It should also assess the data within its established two-year timeline to determine the continued acceptability of pest control products and active ingredients. (1.17–1.20) |
The Agency’s response. Agreed. Since 2008, the Pest Management Regulatory Agency has taken action to limit the number of conditional registrations, which are used only where risks have been deemed acceptable and where additional, confirmatory information has been considered desirable. Currently, one percent of pesticide registrations are conditional in nature; most of these have been conditional for less than five years. Some pesticides have been conditionally registered for longer periods, mainly because the Agency has required information that would take multiple years to generate (for example, new types of scientific studies to be carried out). The Agency believes it could be possible to eliminate the use of conditional registrations and is developing options towards that goal. Stakeholders will be consulted on any proposed change in policy. For the remaining conditional registrations, on a biannual basis, the Agency will post a status report on conditional registrations, including whether the requested information has been received, the actions that have been taken, and the performance in reviewing the data within the two-year timeline. Where necessary, the Agency will use existing authorities in the Pest Control Products Act to ensure that the requested information is provided according to the established timelines. |
Re-evaluations of pesticides
| Recommendation | Response |
|---|---|
|
1.47 The Pest Management Regulatory Agency should finalize and apply the methodology required to ensure that it considers cumulative health effects of pesticides in its re-evaluations, as required by the Pest Control Products Act. (1.29–1.33, 1.40–1.42) |
The Agency’s response. Agreed. The Pest Management Regulatory Agency agrees with the recommendation and the importance of ensuring that the cumulative effects of pesticides are considered where necessary. This is an emerging area in the science used to regulate pesticides, and the Agency is actively working with international partners to understand how to best apply these principles to protect the health and safety of Canadians. Most countries, including Canada, are working on a cumulative risk assessment methodology for pesticides. However, the international scientific community has not yet reached a consensus on how to best assess the cumulative effects of pesticides. The Agency anticipates having its own methodology for cumulative risk assessments in place in the 2017–18 fiscal year. |
|
1.61 The Pest Management Regulatory Agency should establish realistic timelines for completing its re-evaluations of active ingredients registered before 1995, and should complete them accordingly. (1.48–1.60) |
The Agency’s response. Agreed. The Pest Management Regulatory Agency has completed the majority of its re-evaluations of active ingredients registered before 1995 and is committed to completing the remainder of the re-evaluations by 2020. The Agency continues to make progress on the remaining 46 and, where necessary, has implemented interim measures to further protect health and the environment during the review process. The Minister of Health’s multi-stakeholder Pest Management Advisory Council recently supported the Agency’s three-year re-evaluation work plan. The Agency will update the current work plan, in collaboration with the Council, in order to ensure that the remaining 46 re-evaluations are completed by 2020. In completing the re-evaluations, the Agency will collaborate with other foreign regulators, as appropriate, including the US Environmental Protection Agency. The Pest Management Regulatory Agency will continue to report on progress on an annual basis. |
|
1.64 With respect to its overall work planning for re-evaluations, the Pest Management Regulatory Agency should ensure that its schedule of work is complete and up to date. (1.48–1.52, 1.62–1.63) |
The Agency’s response. Agreed. The Pest Management Regulatory Agency will publish updated work plans in early 2016 and update them on a yearly basis. These will include clear expectations for timelines, as well as the expected publication date of the final re-evaluation decision. The Agency’s current approach to re-evaluation favours receipt of new information, including incident reports, from any and all stakeholders even where this material might arrive very late in the review process. While this approach helps ensure re-evaluations are based on the latest and most complete set of information available, it unfortunately often delays the final decision, as the Agency must then reconsider its assessment. To address this challenge and to meet pre-established timelines, the Agency will consult on and develop a policy with clear rules and criteria for submitting information in order to make timely decisions. |
Cancellation of pesticide registrations
| Recommendation | Response |
|---|---|
|
1.84 When the Pest Management Regulatory Agency determines that the risks posed by a pesticide are unacceptable for all uses, it should act promptly to cancel the pesticide’s registration. (1.80–1.83) |
The Agency’s response. Agreed. The Pest Management Regulatory Agency agrees that unacceptable risks presented by pesticides should be addressed promptly. Where unacceptable risks are identified but can be appropriately addressed through mitigation measures, the Agency acts promptly to require such measures. These can range from cancellation of certain uses, to the enhancement of the use of personal protective equipment by workers, to the implementation of additional application restrictions. Where risks are imminent and no appropriate mitigation measures exist, the Agency’s policy is to cancel all uses. In some circumstances where risks are only considered unacceptable in the context of very long-term usage, and where mitigation measures can be put in place as an effective interim solution, the Agency may implement a phase-out period commensurate with the level of risk. This approach ensures the protection of human health and the environment and provides lead time for improved products to be introduced. The use of orderly product phase-outs, where risks can be mitigated in the interim, is a widely accepted practice in use by the US Environmental Protection Agency and other advanced economies. In 2016, to improve transparency, the Agency will consult publicly on its current approach for establishing a phase-out of products (including extensions). |
Communication with the public
| Recommendation | Response |
|---|---|
|
1.93 The Pest Management Regulatory Agency should maintain its public registry of pest control products, as required by the Pest Control Products Act, to clearly communicate which products are conditionally registered and what their associated conditions of registration are. (1.91–1.92) |
The Agency’s response. Agreed. At present, while some information on which products are conditionally registered and their associated conditions is currently available on the Pest Management Regulatory Agency’s section of Health Canada’s website, other information is provided to members of the public upon request. Recognizing the importance of promoting greater transparency in the regulation of pesticides, the Pest Management Regulatory Agency is constantly striving to find ways to more effectively communicate information on conditional registrations with regulated parties, affected stakeholders, and all Canadians. In 2016, all conditions associated with conditional registrations will be published on the Health Canada website. |
|
1.97 To ensure that end users can make informed decisions on pesticide use, the Pest Management Regulatory Agency should consistently communicate timely and readily accessible information on |
The Agency’s response. Agreed. The Pest Management Regulatory Agency acknowledges that the current communication of the information can be improved by clearly identifying which products (rather than active ingredient) are implicated. Recently, the Agency has increased the clarity of its regulatory documents by including timelines for the implementation of regulatory decisions. In 2016, the Agency will identify further opportunities to improve communication with stakeholders and the Canadian public regarding the removal of pesticides or their uses, and the associated timelines. The Agency will also revise decision documents to provide a clear summary of the products it has decided to remove from the market, what uses it has decided to cancel, what mitigation measures it has decided to put in place, and when these changes will take effect. |
PDF Versions
To access the Portable Document Format (PDF) version you must have a PDF reader installed. If you do not already have such a reader, there are numerous PDF readers available for free download or for purchase on the Internet: